The advantages symbionts provide their hosts have influenced the development, ecology, and evolution of eukaryotic life on earth. Yet, we know very little about how and why these associations arose. The overarching goal of my research is to understand the origin of host-microbe associations and how symbiosis shapes host and microbial evolution.
Symbiont-conferred benefits include nutrient provisioning and protection from biotic and abiotic factors
How do novel beneficial associations evolve?
|
The impact of a protective microbe on host adaptation
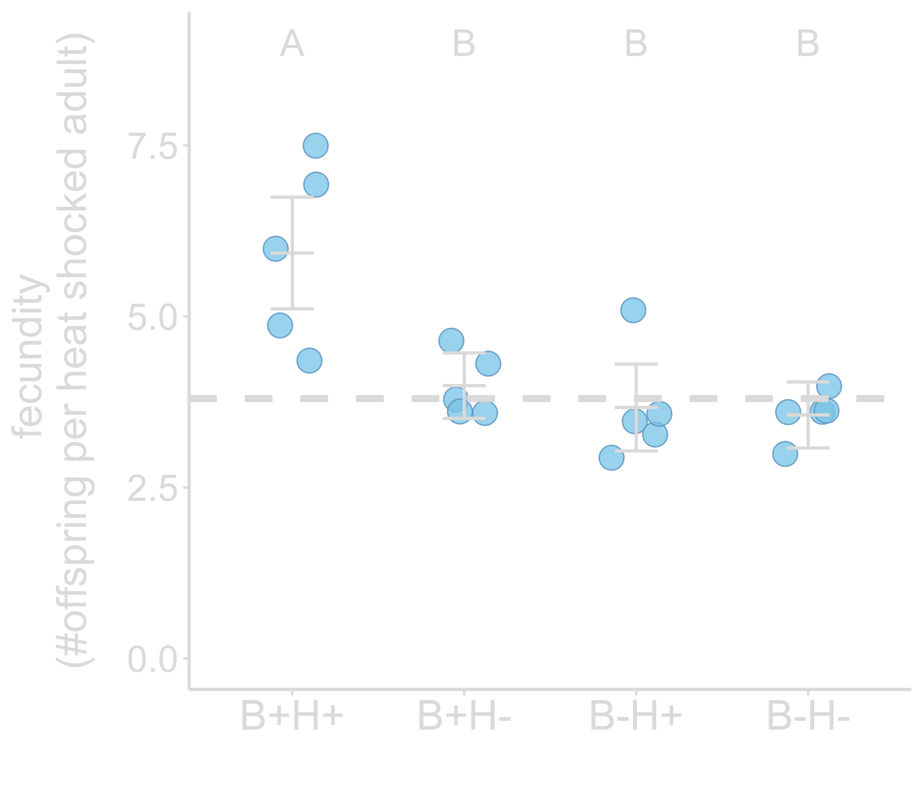
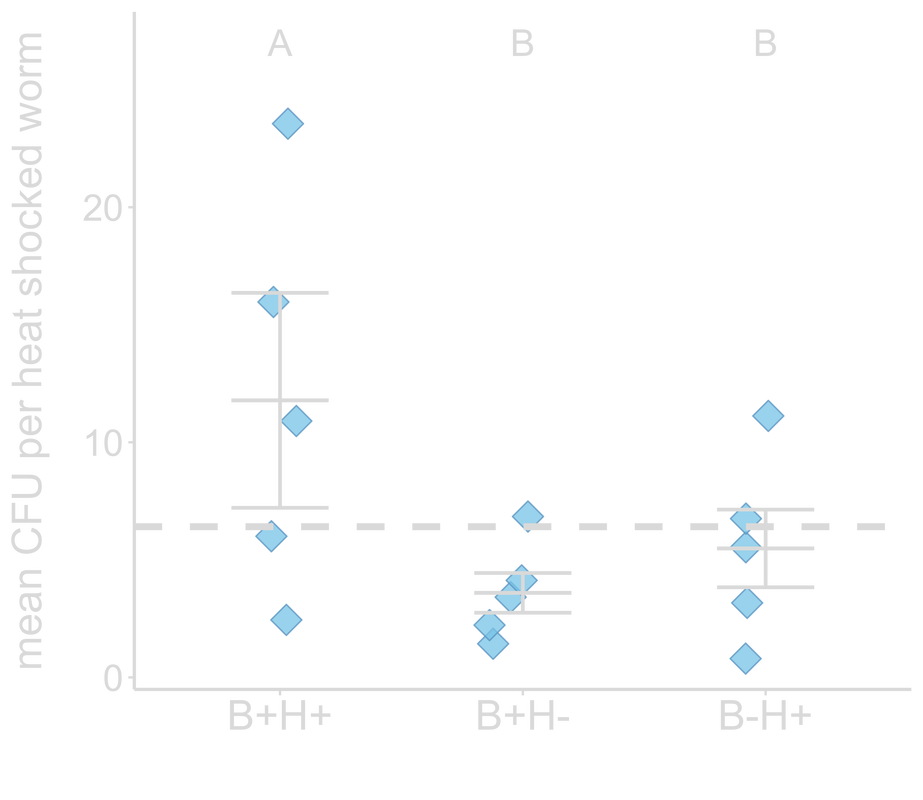
Evolution with a protective bacterium could either enhance or hinder host adaptation, with the former selecting for hosts that harbor the bacterium. During my PhD with Nicole Gerardo and Levi Morran at Emory, we asked what happens when an incipient host is exposed to a protective bacterium under novel environmental conditions? We addressed this question by passaging the nematode Caenorhabditis elegans in the presence of (non-evolving) protective Bacillus subtilis bacterium under heat stress for 20 selection generations. We found that association with novel protective bacteria facilitates host adaptation to stress. Hosts exhibited the greatest fitness gain when evolved in the presence of the bacterium under heat stress, compared to in the absence of the bacterium and/or stress. These hosts also harbored the highest B. subtilis abundance by the end of the experiment. Our findings demonstrate that a bacterium with no evolutionary history with the host facilitated its adaptation, and that host-driven evolution can yield benefits for both host and microbes, potentially leading to symbiosis and even mutualism. Top: Hosts passaged with protective bacteria under heat stress (B+H+) produced the most offspring when heat shocked after generation 20, compared to hosts evolving in the absence of stress (B+H-) or the bacteria (B-H+).
Bottom: Hosts with the most fitness gain (B+H+) also evolved to accommodate more of the bacteria. Dashed line indicates ancestral fitness. From Hoang et al. 2021, Evolution Letters |
|
Coevolution’s role in the establishment of beneficial associations
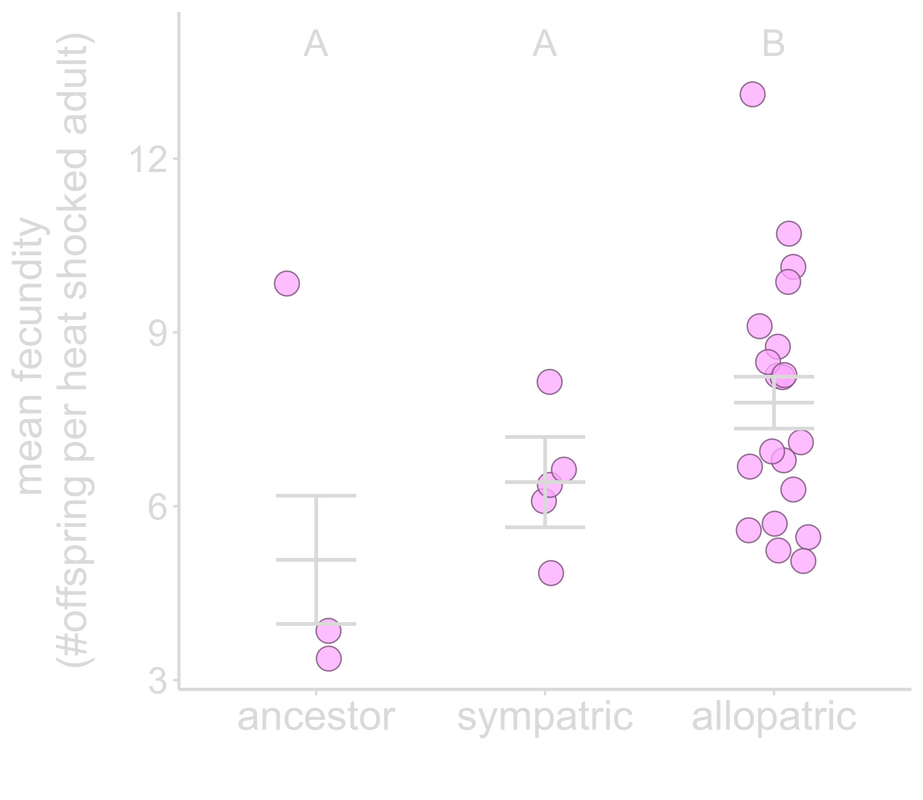
Reciprocal selection between host and microbe drives the evolution of both species. Classical symbiosis models have shown that hosts harboring well-matched symbionts have greater capabilities to thrive in their environments. Yet, despite the benefits hosts gain from harboring symbionts, conflicts can still arise—each partner can act based on their own selfish interests. In new associations, hosts may be prone to exploitation by their symbionts due to the ability of microbes to evolve more rapidly. Continuing with the B. subtilis-C. elegans system, we asked could coevolution alleviate any potential conflicts and increase fitness benefits for hosts, or, alternatively, impede host adaptation and thus the formation of nascent associations? To directly test the role of coevolution in shaping the establishment of symbioses, we co-passaged C. elegans nematodes with B. subtilis bacteria under heat stress for 20 selection generations. Co-passaged hosts did not improve their fitness. Moreover, co-passaged hosts exhibited reduced fitness when paired with their local bacteria compared to bacteria that did not share an evolutionary history with them. Overall, hosts were constrained by their local bacteria and were unable to adapt optimally to stress, countering the expectation that coevolution promotes stability between host and symbiont. Our results highlight the potential underlying conflicts between partners at the onset of an association, particularly those faced by the slower-evolving partner. |
Hosts paired with bacteria with shared evolutionary history produce fewer offspring.
From Hoang et al. 2022, Evolution |
How does symbiosis affect evolutionary trajectories?
Mechanisms of protection employed by defensive symbionts
|
The impact of host-symbiont interaction on pathogen evolution
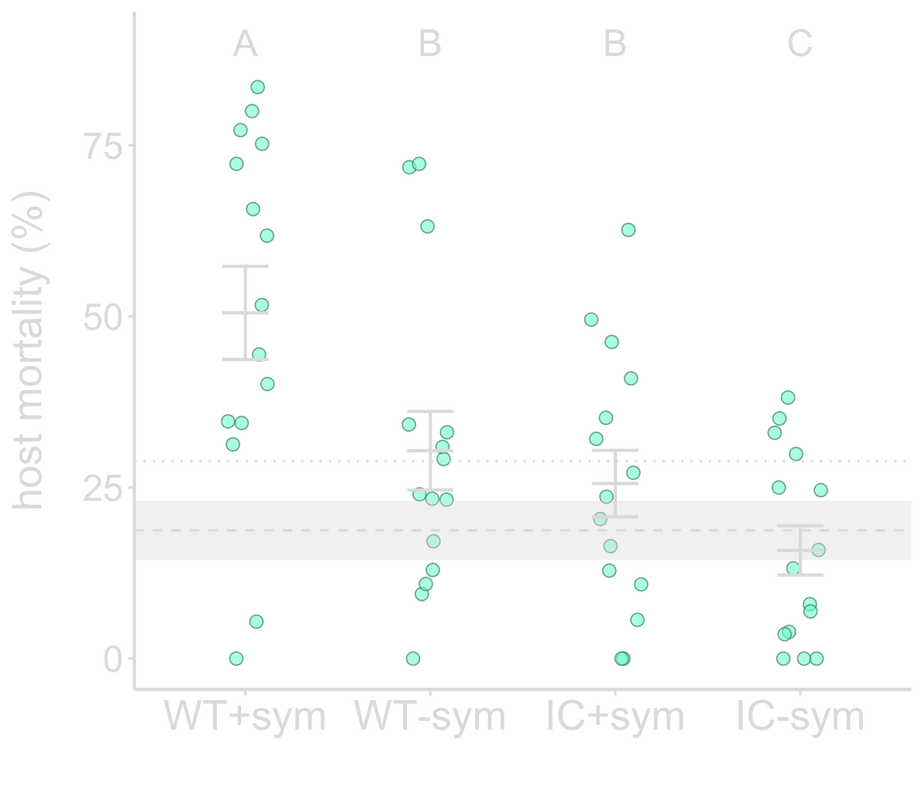
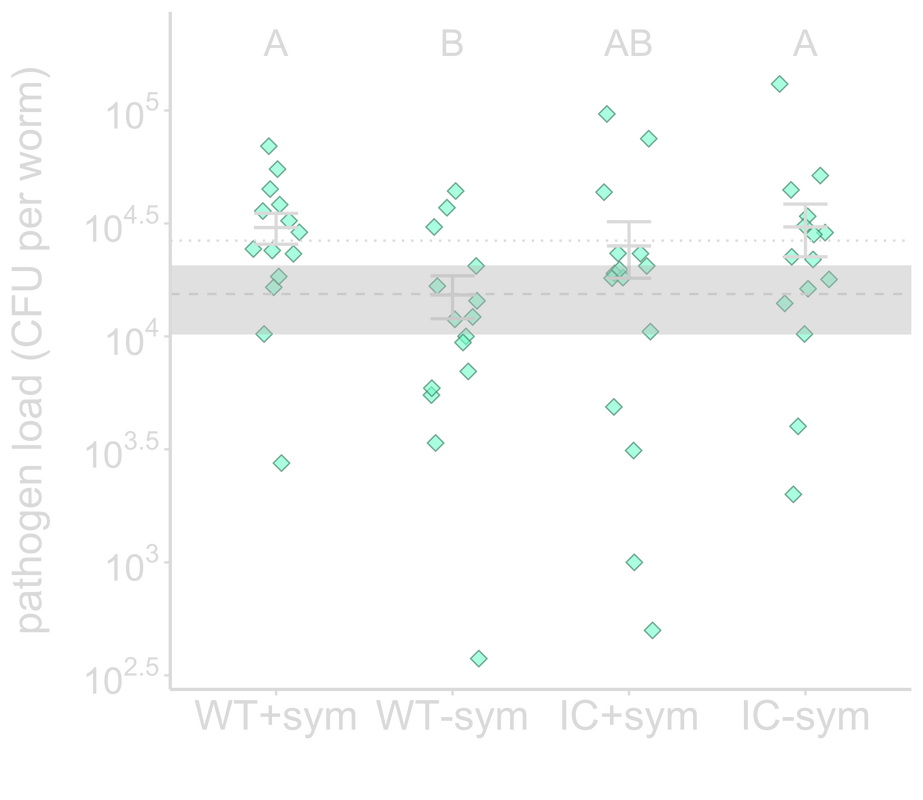
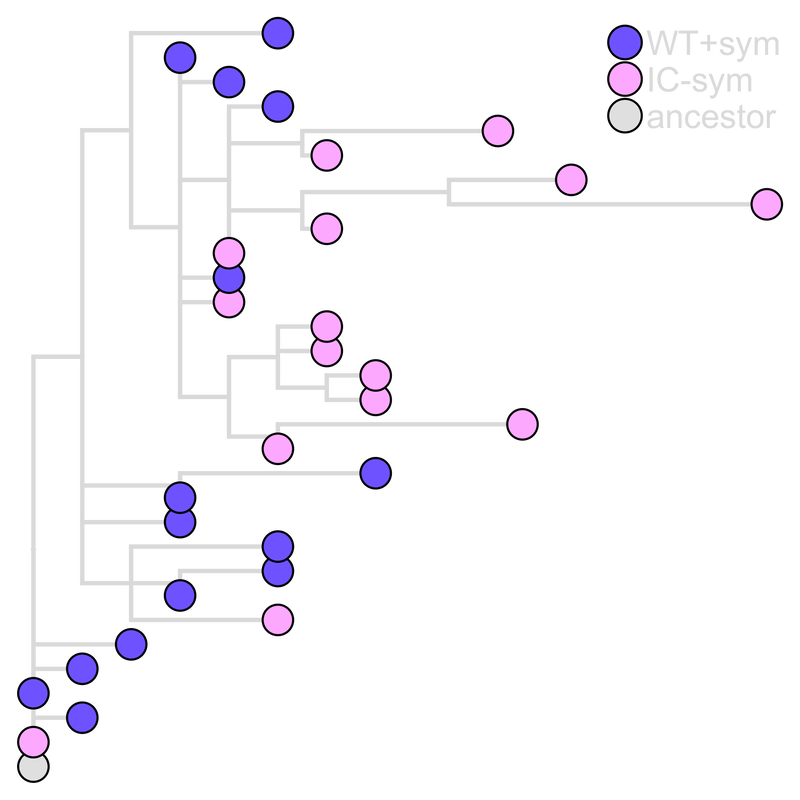
In addition to protection from abiotic stressors, symbionts can defend their hosts against pathogen infection by priming host immunity. For my postdoctoral work with Kayla King at Oxford and Tim Read at Emory, we examined how symbiont-mediated immune priming affects pathogen virulence evolution. We evolved the opportunistic pathogen Pseudomonas aeruginosa through C. elegans nematodes primed by their symbiont, P. mendocina, and demonstrated that symbiont-mediated immune priming selected for increased pathogen virulence. We also found that the most virulent and least virulent populations exhibited the greatest titers within hosts, suggesting that high virulence may not be driven by increased pathogen load. At the genomic level, IC-sym populations are more genetically diverse than WT+sym populations, suggesting that robust host defenses may constrain pathogen molecular evolution. These findings stress the importance of the impact protective symbionts can have on hosts in the long-term, where negative consequences for hosts can arise despite the benefits symbionts provide in the short- term. Top: Pathogen passaged through symbiont-primed hosts (WT+sym) evolved to be the most virulent compared to those passaged through hosts that were not primed (WT-sym), or immunocompromised (IC+sym and IC-sym).
Middle: WT+sym and IC-sym populations had the highest within-host abundance. Dashed line indicates pathogen passaged without host, and dotted line designates the ancestral pathogen. Bottom: IC-sym clones are more genetically distant from the ancestor than WT+sym clones. |